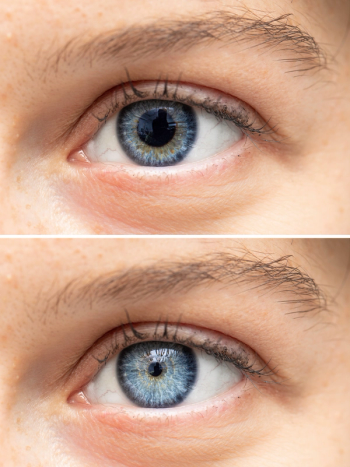
The announcement represents the first FDA-approved, fixed-combination of tropicamide and phenylephrine for mydriasis, according to the company.

The announcement represents the first FDA-approved, fixed-combination of tropicamide and phenylephrine for mydriasis, according to the company.

Ronald Zambrano, lab supervisor for the advanced Ophthalmic Imaging Lab at NYU Langone, talks about lamina cribrosa pressure and predicting structural glaucoma progression at this year's ARVO meeting.

The company’s CT Lucia 621P monofocal IOL enables surgeons to treat a wide range of cataract patients while supporting optimised visual outcomes.

Dr Edmund Arthur shares his research on the early detection of diabetic retinopathy in underserved communities at this year's ARVO meeting.

Dr T.Y. Alvin Liu talks about predictive AI and its uses in ophthalmology and screening of the eyes at this year's ARVO meeting.

Dr Carl Danzig, MD, FASRS, spoke at this year's ARVO meeting on the post hoc analysis for the GATHER trials regarding vision loss and patients receiving ACP 2/mg versus sham.

Dr Caroline Baumal talks about Apellis' presentations focusing on visual function, imaging and artificial intelligence at this year's ARVO meeting.

Dr Deborah Ferrington talks about her ARVO presentation on using human donor tissue to identify the mechanism responsible for the death of the retinal pigment epithelium.

A research team of scientists from a trio of European universities, led by a Northumbria University academic, has received funding to research age-related macular degeneration.

Horus will increase Iluvien’s commercial presence with Nordic retinal specialists.

Angela Carneiro, MD, PhD, discusses her position on a discussion titled Presence of Macular Neovascularization on OCTA is Predictive of Subsequent Exudation with David Hutton, Executive Editor, Ophthalmology Times®.

EMBL-EBI researchers use UK Biobank data to uncover new information about rare diseases of the eye.

Under the terms of the agreement, Grifols to have worldwide exclusive commercial rights to Selagine’s treatment upon regulatory authorization, expected in early 2029.

David S. Boyer, MD, presented data from the PHOTON study examining the safety and efficacy of high-dose aflibercept for treatment of diabetic macular edema at the 2023 Angiogenesis, Exudation, and Degeneration conference.

Arun Singh, MD, discusses Retinoblastoma in 2023 and the advancements of tumor chemotherapy from his presentation at the Asia-Pacific Academy of Ophthalmology (APAO) 2023 conference.

In a presentation at the Angiogenesis, Exudation, and Degeneration 2023 conference, Justis Ehlers, MD, presented "Ellipsoid Zone Integrity in Dry AMD: An Imaging Biomarker for Progression Risk and Clinical Trial Endpoint.”

Rishi Singh, MD, presented data from the Phase 3 DERBY and OAKS clinical trials for geographic atrophy at the 2023 Angiogenesis, Exudation, and Degeneration conference.

According to a news release from Bausch + Lomb and biomedical laser company Modulight, the FDA has approved the ML6710i photodynamic laser for equivalent use with Visudyne.

Pegcetacoplan is the only GA treatment to demonstrate increased effects over 24 months across a broad patient population.

The funding will give a boost to the development of the company's novel photo-crosslinked EyeLief, EyeLief-SD, and OcuLie biodegradable drug-delivery technologies.

Shawn O'Neil of ViaLase shares updates on the company's femtosecond laser, image-guided, high-precision trabeculotomy (FLigHT) treatment.

Adam Szaronos, President and CEO of Trukera Medical, discusses the recent rebranding of TearLab as well as the launch of the ScoutPro Osmolarity System.

At AAO 2022, Dr Mitch Shultz provides discourse on iStent inject vs Hydrus contralateral eye evaluation data.

At the 2022 American Academy of Ophthalmology meeting, Dr Philip Dockery, MPH, presents, "Effectiveness of corneal crosslinking in young patients."

Dr Mark H. Blecher dives into his AAO 2022 topic: "One-Year Visual Outcomes Following Monocular Implantation with a Small Aperture IOL."
David Hutton discusses the AAO 2022 talk, "Corneal Oedema, Opacifications, Ectasia, and Bacterial Keratitis: Diagnosis and Treatment Strategies from the Preferred Practice Pattern Guidelines," with presenter Dr Francis S. Mah.

According to Nicox SA, the patients made their final 3-month visit last week. Top line results of the trial are expected in November.

A team of investigators has found that myopic refractive error is linked with an increased risk of primary open-angle glaucoma, and they indicate that the connection has a genetic foundation.
The companies note that data from Study CPN-302 confirm the results in the first Phase 3 study, CPN-301, and they demonstrate the clear benefits of treating patients after cataract surgery with APP13007.
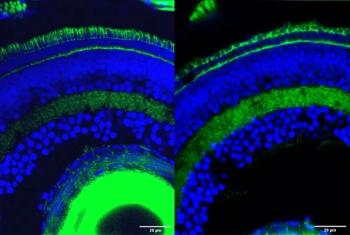
The findings of this study may have revealed the crucial role of gene fragments in retinal development and vision.