Book excerpt: Rescuing failed penetrating keratoplasty grafts
Penetrating keratoplasty remains a popular choice for certain patients
Image used with permission from Springer Nature.

In a new book, Jorge L. Alió, MD, PhD, FEBOphth, and Jorge L. Alió del Barrio, MD, PhD, FEBOS-CR, FWCRS, explore the rapidly changing field of keratoplasty. In this excerpt from the book, our authors discuss protocols for addressing failed grafts.
Modern Keratoplasty: Surgical Techniques and Indications is available as part of the Essentials in Ophthalmology series from Springer Nature.
Introduction
Corneal transplantation is the world’s most frequent type of human tissue graft, with more than 180,000 corneal grafts performed every year worldwide.1 Corneal transplantation aims to restore visual function when it is severely and irreversibly impaired by end-stage corneal disease after conservative surgical or medical treatment options have failed. Corneal transplantation techniques have evolved considerably over the last 2 decades, specifically with regard to lamellar techniques and the optimisation and standardisation of endothelial keratoplasty techniques in particular; however, there remains a critical shortage of corneal graft tissue, and it has been estimated that there is only one corneal donor available for every 70 needed.1 The global shortage of corneal tissue is likely to grow as the cumulative number of patients with transplants increases over time, making corneal graft failure one of the most common indications for corneal transplantation today.2 Corneal graft failure is defined as the failure of the corneal transplant to adequately restore the visual function of the patient in the absence of concomitant pathology.
Primary graft failure is defined as the presence of a diffusely oedematous corneal graft on the first postoperative day that fails to clear at any time postoperatively, with a lack of an identifiable cause of corneal graft failure within 3 months from the transplant.2
Secondary graft failure (SGF) involves the deterioration of vision as a consequence of the loss of corneal transparency in a previously functional corneal graft, usually due to the presence of diffuse and irreversible corneal edema in a previously functional transplant as a consequence of endothelial failure from immunological (rejection) or nonimmunological (chronic late endothelial loss) events.
Morphological graft failure refers to clear grafts without transparency loss but conferring poor visual function as a result of severe irregular astigmatism (such as severe recurrent ectasia). It may be considered a subtype of SGF.
Despite the rise of lamellar keratoplasty techniques, penetrating keratoplasty (PK) remains one of the commonest forms of corneal transplantation and remains the procedure of choice for selected indications.3 Even for an eye that is blind—either from corneal or noncorneal pathology—corneal transplantation may still provide significant benefits in terms of making the eye more comfortable and reducing the risk of infection (although a conjunctival flap may be a more suitable alternative in this situation while not necessarily precluding subsequent corneal transplantation).4 Immunological graft failure may usually be diagnosed with confidence 2 to 3 months after the onset of treatment for endothelial rejection. Rates of reversibility of severe endothelial rejection are between 51% and 64%.5-8
Endothelial keratoplasty might confer a number of advantages and should not be discounted, even in the context of a chronically decompensated PK, because of the capacity for remodeling of any stromal opacification that might take a year or more.9 Consideration should also be given to the surgical aim and whether the existing graft, at its best, ever provided the patient with useful vision. If the graft profile was excellent (low/moderate and regular corneal astigmatism), affording the patient a high level of unaided, spectacle-corrected or contact lens–corrected visual acuity prior to failure, then every effort should be made to maintain this with endothelial keratoplasty. At the other extreme, if the profile of the graft was highly irregular even before failure, and the patient had been unable to tolerate a contact lens to correct for this, then it may be more advantageous to attempt a redo PK and improve on the existing levels of astigmatism.
PK graft failure: incidence and risk factors
The survival rate for PK has been reported recently to be between 52% and 98.8% at 10-year follow-ups.3,10-15 This extended range derives from highly variable methods of data collection and reporting by investigators from different parts of the world.3,16 Furthermore, datasets published before the advent of endothelial keratoplasty likely provide a more comprehensive overview of the risks and benefits of repeat PK. PK is now less commonly performed for low-risk indications such as keratoconus or Fuchs dystrophy, where lamellar techniques are now generally regarded as the gold standard. PK is increasingly being reverted to in more complex cases where the technical difficulty inherent to lamellar techniques preclude their success.17
Outcomes with PK are strongly related to the primary diagnosis—the indication for transplantation being the strongest predictor of survival.18 Transplantations performed for keratoconus and stromal dystrophies have a better prognosis than grafts performed for endothelial diseases, bullous keratopathy and postinfective corneal scars, for example.15, 17, 19 Moreover, when a PK is performed for therapeutic and tectonic indications, as opposed to optical rehabilitation, the prognosis is even poorer.13,20 Other wellestablished risk factors for PK failure include a history of previous failed PK, the presence of active ocular inflammation, corneal neovascularisation, other ocular comorbidities, aphakia, larger-graft diameter and the undertaking of combined surgical procedures.15, 18, 21-25
Repeat PK
As a consequence of the recent introduction of posterior lamellar grafts, repeat PK remains the only surgical option for PK grafts that have failed and where the functional potential of the existing transplant has been fundamentally compromised (eg, where there is severe stromal opacification in addition to endothelial dysfunction).
Figure 1. Slit lamp picture and anterior segment OCT image (Visante, Zeiss, Germany) of a patient that received Descemet's stripping DSAEK under a previously failed PK.
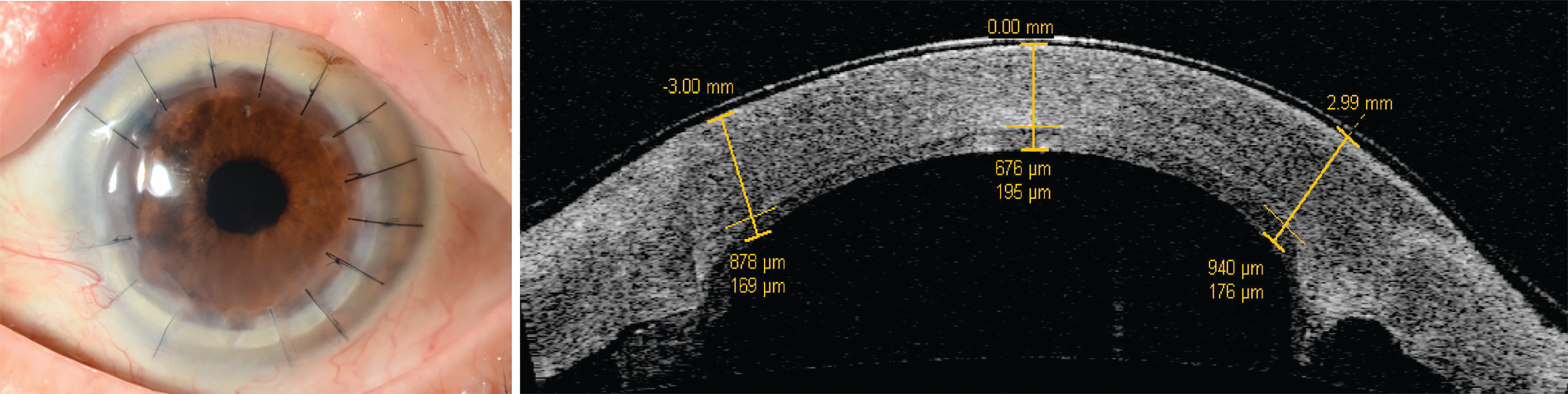
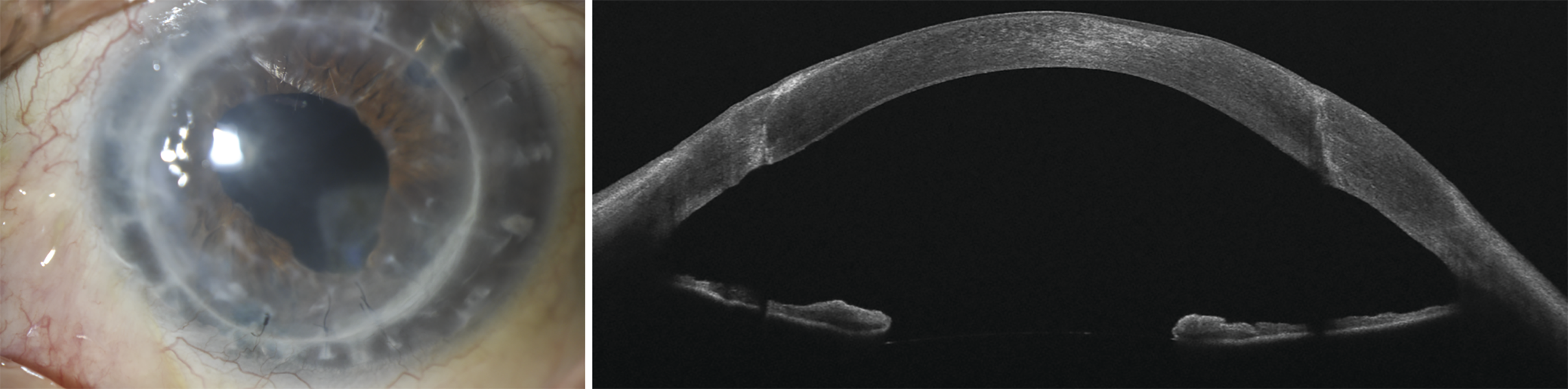
Figure 2. Slit lamp picture and anterior segment OCT image (MS-39, CSO, Italy) of a patient that received non-Descemet stripping DMEK under a previously failed PK. Images are taken 2 years after surgery. The inferior peripheral scar appeared as a consequence of microbial keratitis 1 year after surgery. (Images courtesy of the authors, published with permission from Springer Nature)
While there has been a progressive reduction in the volumes of primary PKs undertaken, the number of PKs constituting regrafts has remained relatively stable in recent years, accounting for 11.3% to 19.0% of the total PKs performed in the United States from 2005 to 2019.26,27 Mattahei and coworkers, in a systematic review of the global indications for PK surgery, found that between 1980 and 2014, PK regrafts represented 9.3% to 18.8% of the total number of PKs performed in developed countries.16
Subsequent PKs usually carry all the risks of the first transplant plus the increased risk of allograft rejection, as well as cumulative comorbidities such as glaucoma.22,28 Consequently, every effort should be made to optimise the condition of the ocular surface, control intraocular pressure, address inflammation (including systemic inflammatory diseases) and manage other pathologies that might impact the risk of rejection such as corneal neovascularisation and herpetic eye disease.22,29 Preoperative imaging, including anterior segment optical coherence tomography, may be helpful in anticipating and planning for additional procedures that may be indicated more frequently with repeat transplantation, including synechiolysis, iridectomy and pupilloplasty.30 For grafts at high risk of rejection—usually defined as one where there are two or more quadrants of neovascularisation or where previous rejection has occurred—there is limited evidence to support the use of both pre- and postoperative systemic immunosuppression.31 Management varies widely, but presurgical systemic corticosteroid treatment improves corneal transplant survival in murine models, and courses of pre- and postoperative oral prednisone (1 mg/kg) or intravenous methylprednisolone are often prescribed in this context, usually in combination with topical steroid treatment and, less often, steroid-sparing agents such as mycophenolate mofetil, cyclosporin A, tacrolimus, sirolimus and topical cyclosporin.6,31-33
A significant proportion of surgeons in one study prescribe topical steroids for use indefinitely in high-risk cases, and oral acyclovir for a median of 6 months in those cases involving Herpes simplex keratopathy.33 There is moderate evidence for the benefit of mycophenolate mofetil on corneal transplant survival, but evidence for the effectiveness of other noncorticosteroid immunosuppressive agents is less conclusive.31,34,35 The case for human leukocyte antigen matching of tissue to the recipient has yet to be demonstrated in studies determining whether this results in a reduction in the risk of allograft rejection compared with random tissue allocation; equally, there is some evidence to suggest a role for the indirect presentation of minor histocompatibility antigens (such as male H-Y antigen) in modulating the risk of corneal transplant rejection and graft survival, but this has yet to inform practice widely, and the effect may not be significant in high-risk cases.36,37
Given the risks of allograft rejection in eyes that have undergone multiple PKs, these may ultimately be better candidates for a keratoprosthesis, such as the Boston type 2 keratoprosthesis (BKPro), if the ocular surface is not compromised by dryness or a lack of blinking.28 Refinements to the device itself and the management of eyes implanted with it have led to an expansion of the indications for the BKPro. There is some evidence to support the use of the BKPro earlier in the pathway of eyes at risk of further failed PKs, with data from one study demonstrating the likelihood of maintaining 20/200 or better vision at 2 years and a clear graft at 5 years was significantly higher with the BKPro than PK in eyes with a previously failed PK (without a higher risk of postoperative glaucoma).38-40
Surgical considerations
Particular care in surgical planning should be undertaken when the original transplant was a therapeutic keratoplasty for microbial keratitis, because disruption of the anterior segment anatomy, eccentrically sited transplants, and comorbidities such as glaucoma and cataract are more common in such cases.41,42 With regard to host trephination, consideration should be given to the indications for the transplant and the reasons for failure. The capacity to heal varies considerably between patients and eyes, and this is further affected by the duration of any steroid treatment and even whether the wound interface is that of a primary PK or a regraft, with evidence for a stronger wound-healing response in the latter.43 It might be assumed, at the point where one might consider suture removal (usually 1 year after surgery), that the graft-host interface is sufficiently healed as to make re-trephination a more obvious choice but, in fact, the strength of the graft-host interface is never much more than that afforded by the endothelium and epithelium—this is what makes PKs particularly vulnerable to traumatic injury and, while most traumatic wound dehiscence occurs within 18 to 24 months of transplantation, this can occur even decades after the surgery.44-46 Consequently, most PKs may be replaced by simply accessing the graft-host junction, peeling out the original graft, and suturing in a transplant of the same diameter.46,47 The appearance on slit lamp examination preoperatively of vessels and stromal opacification at the graft-host interface may give some indication as to the degree of healing that has already taken place and, if significant, a little more resis- tance might be expected with this approach, but remains the favored option in most cases.
As a general rule, the transplant should be as ‘large as possible and as small as necessary’—that is to say, a balance should be struck, in terms of the optical advantages conferred by a larger graft and the reduced risks of allograft rejection associated with smaller diameter transplants.17,18 While peeling out the original donor is relatively easily performed, repeat PK does afford the opportunity to excise the old transplant together with a rim of host cornea by performing a slightly larger trephination—if this can be accomplished while maintaining sufficient clearance from the limbus, ideally at least 1.5 mm. This may be preferred if decentration, high levels of astigmatism or irregularity have been a problem with the original transplant. Otherwise, greater weight should be afforded to the risks of allograft rejection in the context of a repeat PK.28 A ‘peephole’ transplant, sited within a larger-diameter, failed transplant, may offer considerable functional benefit while minimising the risks of further failure, albeit with a flatter profile and higher levels of irregular astigmatism.48 Conversely, a graft that has failed due to infection or a corneal melting disorder may require excision of diseased or thinned tissue far outside of the existing transplant, sometimes up to the limbus—with the caveat that it is almost always worth considering a tectonic anterior lamellar keratoplasty in an acute setting rather than a PK.20,49
Where a large-diameter keratoplasty, such as a total corneal transplant, is indicated, every effort should be made to ensure that the underlying cause has been managed appropriately, that inflammation has settled and that the host limbal stem cell populations are spared by undermining and retracting the corneal limbus prior to trephination.20,50 Some vacuum trephines may not be available in the size required or even suitable for application in these circumstances—in which case marking of the surface, or partial trephination, with a hand-held trephine, followed by a freehand dissection, may permit more controlled excision of the host button.49
Outcomes
Repeat PKs have a higher risk of failure compared with primary PK, with a worse prognosis and poorer visual outcomes. The 5-year graft failure rate for repeat PK is between 34% and 70%,41,53-56 with numerous factors playing a role in the survival of the transplant. Repeated PKs share the same risk factors as primary PK, but there is a higher risk of immunological allograft rejection associated with violation of the immune-privileged status of the cornea. This risk increases with the number of prior corneal grafts performed, especially if they have failed as a result of allograft rejection.25,53,57 In addition, the host corneal bed is usually suboptimal, with high rates of altered iridocorneal angle anatomy, peripheral anterior synechiae and corneal surface disease. When compared with patients receiving primary PK, patients receiving PK regraft are slightly older, have higher use of IOP-lowering drugs and are more prone to develop postoperative corneal neovascularisation, all of which are associated with poor graft survival.56 Finally, as for primary PK, the risk of a failed graft is closely associated with the original graft indication, with the best rates of survival reported for eyes with a primary diagnosis of keratoconus and stromal dystrophies.54
With regard to visual outcomes, repeat PK is generally effective in improving the preoperative best-corrected visual acuity (BCVA), but the results achieved are usually worse than those for primary PK. In fact, a BCVA of 20/40 or better has been reported as comprising only 4.8% to 43.1% of clear PK regrafts at the last follow-up visit.54,58-65 In addition, the standard of final visual acuity achieved is inversely proportional to the number of corneal regrafts performed.59 Despite this, a recent meta-analysis performed by Wang and coworkers found that visual outcomes in PK regrafts were not inferior to the ones for secondary endothelial grafts.66
References
Gain P, Jullienne R, He Z, et al. Global survey of corneal transplantation and eye banking. JAMA Ophthalmol. 2016;134(2):167-173. doi:10.1001/jamaophthalmol.2015.4776
Alio JL, Montesel A, El Sayyad F, Barraquer RI, ArnalichMontiel F, Alio del Barrio JL. Corneal graft failure: anupdate. Br J Ophthalmol. 2020;105(8):1049-1058. doi:10.1136/bjophthalmol-2020-316705
Patel SY. Graft survival after penetrating keratoplasty. Am J Ophthalmol. 2011;151(3):397-398.doi:10.1016/j.ajo.2010.10.006
Alino AM,PerryHD, Kanellopoulos AJ,Donnenfeld ED, Rahn EK. Conjunctival flaps.Ophthalmology. 1998;105(6):1120-1123. doi:10.1016/S0161-6420(98)96017-1
Qazi Y, Hamrah P. Corneal allograft rejection: immunopathogenesis to therapeutics. J Clin Cell Immunol. 2013;2013(9):6. doi:10.4172/2155-9899.S9-006
Kharod-Dholakia B, Randleman JB, Bromley JG, Stulting RD. Prevention and treatment of corneal graft rejection: current practice patterns of the CorneaSociety(2011).Cornea.2015;34(6):609-614. doi:10.1097/ICO.0000000000000403
Hudde T, Minassian DC, Larkin DF. Randomised controlled trial of corticosteroid regimens in endothelial corneal allograft rejection. BrJ Ophthalmol. 1999;83(12):1348-52.
Yamazoe K, Yamazoe K, Shimazaki-Den S, Shimazaki J. Prognostic factors for corneal graft recovery after severe corneal graft rejection following penetrating keratoplasty. BMCOphthalmol. 2013;13:5. doi:10.1186/1471-2415-13-5
Pasari A, Price MO, Feng MT, Price FW Jr. Descemet membrane endothelial keratoplasty for failedpenetrating keratoplasty:visualoutcomesand graft survival. Cornea. 2018;38(2):151-156. doi:10.1097/ICO.0000000000001763
Borderie VM, Boëlle PY, Touzeau O,Allouch C, Boutboul S, Laroche L. Predicted long-term outcome of corneal transplantation. Ophthalmology. 2009;116(12):2354-2360. doi:10.1016/j.ophtha.2009.05.009
FukuokaS,HondaN,OnoK,MimuraT,Usui T, Amano S. Extended long-term results of penetrating keratoplasty for keratoconus. Cornea. 2010;29(5):528-530. doi:10.1097/ICO.0b013e3181c29705
Writing Committee for the Cornea Donor Study Research Group, Mannis MJ, Holland EJ, et al.The effect of donor age on penetrating keratoplasty for endothelial disease: graft survival after 10 years in the Cornea Donor Study. Ophthalmology. 2013;120(12):2419-2427. doi:10.1016/j.ophtha.2013.08.026
TanDTH,JanardhananP,Zhou H,et al. Penetrating keratoplasty in asian eyes: the Singapore Corneal Transplant Study. Ophthalmology.2008;115(6):975-982.e1. doi:10.1016/j.ophtha.2007.08.049
ThompsonRW,PriceMO,BowersPJ,Price FW Jr. Long-term graft survival after penetrating keratoplasty. Ophthalmology. 2003;110(7):1396-1402. doi:10.1016/S0161-6420(03)00463-9
Williams KA, Lowe M, Bartlett C, Kelly TL, Coster DJ; All Contributors. Risk factors for human corneal graft failure within the Australian corneal graft registry. Transplantation.2008;86(12):1720-1724. doi:10.1097/TP.0b013e3181903b0a
Matthaei M, Sandhaeger H, Hermel M, etal.Changing indications in penetrating keratoplasty: a systematic review of 34 years of global reporting.Transplantation. 2017;101(6):1387-1399. doi:10.1097/TP.0000000000001281
Williams KA, Keane MC, Coffey NE,Jones VJ, Mills RAD, Coster DJ.TheAustralian Corneal Graft Registry: 2018 Report. Flinders University/South Australian Health and Medical Research Institute;2018.
Barraquer RI, Pareja-Aricò L, Gómez-Benlloch A, Michael R. Risk factors for graft failure after penetrating keratoplasty. Medicine (Baltimore).2019;98(17):el5274. doi:10.1097/MD.0000000000015274
WilliamsKA, EstermanAJ, BartlettC, Holland H, Hornsby NB, Coster DJ. How effective is penetrating corneal transplantation? Factors influencing long-term outcome in multivariate analysis. Transplantation. 2006;81(6):896-901. doi:10.1097/01.tp.0000185197.37824.35
Ang M, Mehta JS, Sng CCA, Htoon HM, Tan DTH. Indications, outcomes, and risk factors for failure in tectonic keratoplasty. Ophthalmology. 2012;119(7):1311-1319. doi:10.1016/j.ophtha.2012.01.021
Perera C, Jhanji V, Lamoureux E, Pollock G, Favilla I, Vajpayee RB. Clinical presentation, risk factors and treatment outcomes of first allograft: rejection after penetrating keratoplasty in early and late postoperative period. Eye (Lond). 2012;26(5):711-717. doi:10.1038/eye.2012.14
Price MO, Thompson RW Jr, Price FW Jr. Risk factors for various causes of failure in initial corneal grafts. Arch Ophthalmol. 2003;121(8):1087-1092. doi:10.1001/archopht.121.8.1087
Writing Committee for the Cornea Donor Study Research Group, Sugar A, Gal RL, et al. Factors associated with corneal graft survival in the cornea donor study. JAMA Ophthalmol. 2015;133(3):246-254. doi:10.1001/jamaophthalmol.2014.3923
Sugar A, Tanner JP, Dontchev M, et al; Cornea Donor Study Investigator Group. Recipientrisk factors for graft failure in the cornea donor study. Ophthalmology.2009;116(6):1023-1028. doi:10.1016/j.ophtha.2008.12.050
Wilson SE, Kaufman HE. Graft failure after penetrating keratoplasty. Surv Ophthalmol. 1990;34(5):325-356. doi:10.1016/0039-6257(90)90110-h
Park CY, Lee JK, Gore PK, Lim CY, Chuck RS. Keratoplasty in the United States: a 10-year review from 2005 through 2014. Ophthalmology. 2015;122(12):2432-2442. doi:10.1016/j.ophtha.2015.08.017
2019 Eye Banking Statistical Report. Eye Bank Association of America; 2019.
Maguire MG, Stark WJ, Gottsch JD, et al. Risk factors for corneal graft failure and rejection in the collaborative corneal transplantation studies. Collaborative Corneal Transplantation Studies Research Group. Ophthalmology. 1994;101(9):1536-1547. doi:10.1016/s0161-6420(94)31138-9
Hos D, Le VNH, Hellmich M, et al. Risk of corneal graft rejection after high-risk keratoplasty following fine-needle vessel coagulation of corneal neovascularization combined with bevacizumab: a pilot study. Transplant Direct. 2019;5(5):e452. doi:10.1097/TXD.0000000000000894
Ang M, Baskaran M, Werkmeister RM, et al. Anterior segment optical coherence tomography. ProgRetinEye Res. 2018;66:132-156. doi:10.1016/j.preteyeres.2018.04.002
Abudou M, Wu T, Evans JR, Chen X. Immunosuppressants for the prophylaxis of corneal graft rejection after penetrating keratoplasty. Cochrane Database Syst Rev. 2015;2015(8):CD007603. doi:10.1002/14651858.CD007603.pub2
Kim HK, Choi JA, Uehara H, Zhang X, Ambati BK, Cho YK. Presurgical corticosteroid treatment improves corneal transplant survival in mice. Cornea. 2013;32(12): 1591-1598. doi:10.1097/ICO.0b013e31829ebb0d
Koay PY, Lee WH, Figueiredo FC. Opinions on risk factors and management of corneal graft rejection in the United Kingdom. Cornea. 2005;24(3):292-296. doi:10.1097/01.ico.0000138841.44926.f8
Birnbaum F, Mayweg S, Reis A,et al. Mycophenolate mofetil (MMF) following penetrating high-risk keratoplasty: long-term results of a prospective, randomised, multicentre study. Eye (Lond). 2009;23(11):2063-2070. doi: 10.1038/eye.2008.402.
Sinha R, Jhanji V, Verma K, Sharma N, Biswas NR, Vajpayee RB. Efficacy of topical cyclosporine A2% in prevention of graft rejection in high-risk keratoplasty:a randomized controlled trial. Graefes Arch Clin Exp Ophthalmol. 2010;248(8):1167-1172. doi: 10.1007/s00417-010-1388-8.
Bohringer D, Grotejohann B, Ihorst G, Reinshagen H, Spierings E, Reinhard T. Rejection prophylaxis in corneal transplant. Dtsch Arzteblatt Int. 2018;115(15):259-265. doi: 10.3238/arztebl.2018.0259.
Hopkinson CL, Romano V, Kaye RA, et al. The influence of donor and recipient gender incompatibility on corneal transplant rejection and failure. Am J Transplant. 2017;17(1):210-217. doi: 10.1111/ajt.13926
Hager JL, Phillips DL, Goins KM, et al.Boston type 1 keratoprosthesis forfailed keratoplasty.Int Ophthalmol. 2016;36(1):73-78. doi: 10.1007/s10792-015-0078-2
Ahmad S, Mathews PM, Lindsley K, et al. Boston type I keratoprosthesis versus repeat donor keratoplasty for corneal graft failure: a systematic review and meta-analysis. Ophthalmology.2016;123(1):165-177. doi: 10.1016/j.ophtha.2015.09.028
Fadous R, Levallois-Gignac S, Vaillancourt L, Robert MC, Harissi-Dagher M. The Boston keratoprosthesis type 1 as primary penetrating corneal procedure. Br J Ophthalmol.2015;99(12):1664-668. doi:10.1136/bjophthalmol-2014-306161
RamamurthyS, ReddyJC, VaddavalliPK, Ali MH, Garg P. Outcomes of repeat keratoplasty for failed therapeutic keratoplasty. Am J Ophthalmol. 2016;162(83-8):e2. doi: 10.1016/j.ajo.2015.11.004.
SrujanaD,KaurM,UrkudeJ,RathiA,Sharma N,TitiyalJS.Long-termfunctionalandanatomicoutcomesofrepeatgraftafteroptically failed therapeutic keratoplasty. Am J Ophthalmol. 2018;189:166-175. doi: 10.1016/j.ajo.2018.03.011
Abdelkader A. Changes in corneal wound healing and graft biomechanics after primary penetrating keratoplasty versus repeat penetrating keratoplasty. Cornea. 2019;38(8):1006-1010. doi:10.1097/ICO.0000000000001928
Raber IM, Arentsen JJ, Laibson PR. Traumatic wound dehiscence after penetrating keratoplasty. Arch Ophthalmol. 1980;98(8):1407-1409. doi:10.1001/archopht.1980.01020040259009
BinderPS,AbelRJr,PolackFM,KaufmanHE. Keratoplasty wound separations. Am J Ophthalmol. 1975;80(1):109-115. doi:10.1016/0002-9394(75)90880-6
Pettinelli DJ, Starr CE, Stark WJ. Late traumatic corneal wound dehiscence after penetrating keratoplasty. Arch Ophthalmol. 2005;123(6):853-856. doi:10.1001/archopht.123.6.853
Kirkness CM, Ezra E, Rice NS, Steele AD. The success and survival of repeat corneal grafts. Eye. 1990;4(1):58-64. doi:10.1038/eye.1990.6
Seitz B, Langenbucher A, Kuchle M, NaumannGO. Impact of graft diameter on corneal power and the regularity ofpostkeratoplastyastigmatism before and after suture removal. Ophthalmology. 2003;110(11):2162-2167. doi:10.1016/S0161-6420(03)00659-6
Bessant DA, Dart JK. Lamellar keratoplasty in the managementofinflammatorycornealulcerationand perforation.Eye. 1994;8(1):22-28. doi:10.1038/eye.1994.4
Jones DH, KirknessCM. A new surgical technique for keratoglobus-tectonic lamellar keratoplasty followed by secondary penetrating keratoplasty. Cornea.2001;20(8):885-887. doi:10.1097/00003226-200111000-00022
Lee RM, Lam FC, Georgiou T, Paul B, Then KY, Mavrikakis I, et al. Suturing techniques and postoperativemanagementinpenetratingkeratoplasty in the United Kingdom. Clin Ophthalmol. 2012;6:1335-1340. doi:10.2147/OPTH.S35460
Koenig SB, Harris GJ. Temporary suture tarsorrhaphy after penetrating keratoplasty. Cornea. 1991;10(2):121-122. doi:10.1097/00003226-199103000-00005
Aboshiha J, Jones MNA, Hopkinson CL, Larkin DFP.Differential survival ofpenetrating andlamellar transplants in management of failed corneal grafts. JAMA Ophthalmol. 2018;136(8):859-865. doi:10.1001/jamaophthalmol.2018.1515
Al-Mezaine H, Wagoner MD. Repeat penetrating keratoplasty: indications, graft survival, and visual outcome. Br J Ophthalmol.2006;90(3):324-327. doi:10.1136/bjo.2005.079624
Kitazawa K, Wakimasu K, Kayukawa K, et al. Moderately long-term safety and efficacy of repeat penetrating keratoplasty. Cornea. 2018;37(10):255-259. doi:10.1097/ICO.0000000000001694
Weisbrod DJ, Sit M, Naor J, Slomovic AR. Outcomes of repeat penetrating keratoplasty and risk factors for graft failure. Cornea. 2003;22(5):429-434. doi:10.1097/00003226-200307000-00008
Jabbehdari S, Rafii AB, Yazdanpanah G, HmmahP, Holland EJ, Djalilian AR. Update on the management of high-risk penetrating keratoplasty.Curr Ophthalmol Rep. 2017;5(1):38-48. doi:10.1007/s40135-017-0119-2
Beckingsale P, Mavrikakis I, Al-Yousuf N, Mavrikakis E, Daya SM. Penetrating keratoplasty: outcomes from acorneal unit compared to national data. Br J Ophthalmol. 2006;90(6):728-731. doi:10.1136/bjo.2005.086272
Bersudsky V, Blum-Hareuveni T, Rehany U, Rumelt S. The profile of repeated corneal transplantation. Ophthalmology. 2001;108(3):461-469. doi:10.1016/s0161-6420(00)00544-3
Keane MC, Galettis RA, Mills RAD, Coster DJ, Williams KA. A comparison of endothelial and penetrating keratoplasty outcomes following failed penetrating keratoplasty: a registry study. Br J Ophthalmol. 2016;100(11):1569-1575. doi:10.1136/bjophthalmol-2015-307792
Kitzmann AS, Wandling GR, Sutphin JE, Goins KM, Wagoner MD. Comparison of outcomes of penetrating keratoplasty versus Descemet's stripping automated endothelial keratoplasty for penetrating keratoplasty graft failure due to corneal edema. Int Ophthalmol. 2012;32(1):15-23. doi:10.1007/s10792-012-9518-4
Patel NP, Kim T, Rapuano CJ, Cohen EJ, Laibson PR. Indications for and outcomes of repeat penetrating keratoplasty, 1989-1995. Ophthalmology. 2000;107(4):719-724. doi:10.1016/s0161-6420(00)00003-8
Rapuano CJ, Cohen EJ, Brady SE, Arentsen JJ, LaibsonPR.Indicationsforandoutcomesof repeat penetrating keratoplasty. Am J Ophthalmol. 1990;109(6):689-695. doi: 10.1016/s0002-9394(14)72437-7
Yalniz-Akkaya Z, Nurozler AB, Yildiz EH, OnatM, Budak K, Duman S.Repeat penetrating keratoplasty:indications and prognosis, 1995-2005. EurJ Ophthalmol. 2009;19(3):362-368. doi: 10.1177/112067210901900306.
Barut Seiver O, Karaca I,Palamar M, Egrilmez S, Yagci A. Graft failure and repeat penetrating keratoplasty. ExpClin Transplant. 2021;19(1):72-76. doi:10.6002/ect.2017.0165
Wang F, Zhang T, Kang YW, He JL, Li SM, LiSW.Endothelialkeratoplastyversusrepeatpenetratingkeratoplastyafterfailedpenetratingkeratoplasty: asystematic review and meta-analysis.PLoS ONE. 2017;12(7):1-11. doi:10.1371/journal.pone.0180468. eCollection 2017.

Newsletter
Join ophthalmologists across Europe—sign up for exclusive updates and innovations in surgical techniques and clinical care.