
Cataract surgery in Cambodia – Achieving quality outcomes in a challenging setting with the CT LUCIA 621P
Cataract remains a leading cause of visual impairment and blindness globally with the highest rates occurring in low- and middle-income countries. Services provided by volunteer Western ophthalmologists working through non-governmental organizations (NGOs) have an important role in alleviating this burden. Visual acuity is used as the metric for measuring benefit of cataract surgery.
According to guidelines from the World Health Organization (WHO) for monitoring cataract surgery outcomes, “good” BCVA (i.e, 20/20 to 20/60) should be achieved with available and best correction in ≥80% and ≥90% of eyes, respectively.1 Visual acuity after cataract surgery depends in part on the quality of the operation, but also on the quality and long-term performance of the intraocular lens (IOL) chosen for visual rehabilitation.
We regularly volunteer for international missions to Cambodia through the Khmer Sight Foundation NGO and the Augenärzte für die Welt gGmbH NGO to perform cataract surgery and train local ophthalmologists.For an upcoming trip scheduled in the spring of 2022, we agreed to conduct a study sponsored by Carl Zeiss Meditec AG to evaluate outcomes of surgery using the CT LUCIA 621P (Carl Zeiss Meditec, AG) as the implant and the IOLMaster 700 (Carl Zeiss Meditec, AG) for biometry and IOL power calculations.
Because preoperative biometry done in NGO settings are usually performed by A-scan, which is both time consuming and cumbersome, we welcomed the opportunity to obtain accurate preoperative measurements efficiently with the IOLMaster 700. Use of the CT LUCIA 621P was also appealing from a workflow perspective and for safety reasons because this IOL comes fully preloaded in a proprietary single-use injection system (BLUESERT™). In addition, being familiar with the CT LUCIA 621P from experience with it in our private practices, we believed that it offered a number of characteristics that would make it a very attractive choice in the setting of a developing country where continuity of care can be poor so that biocompatibility, resistance to the development of posterior capsule opacification (PCO), and positional stability are of heightened importance for providing durably good visual outcomes.
Features of the CT LUCIA 621P
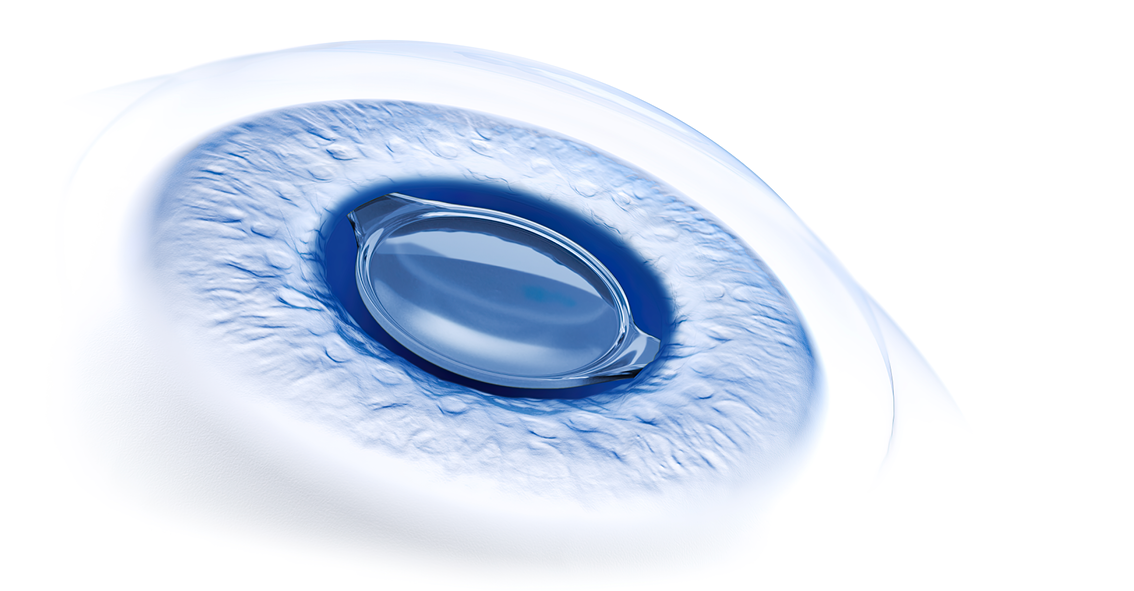
The CT LUCIA 621P is a single-piece, monofocal, aspheric IOL made of a hydrophobic acrylic with a heparin-coated surface*. It measures 13.0 mm in total diameter, has a 6.0 mm optic with a sharp posterior edge and unique patented aspheric design (ZEISS Optic [ZO]), and features step-vaulted C-loop haptics.2,3
Several features of the CT LUCIA 621P confer resistance to PCO development, which is a particularly important benefit for an IOL used in developing countries where patients may be unlikely to have access to Nd:YAG laser capsulotomy. Not only does the sharp posterior optic edge serve as a mechanical barrier to lens epithelial cell migration, but the rigid step-vaulted haptics of the CT LUCIA 621P help to maintain contact between the sharp edge and the posterior capsule at the optic-haptic junction, a site considered as a possible Achilles’ heel in blocking lens epithelial ingrowth.2 Its hydrophobic acrylic material adds value for PCO resistance.5 The C-loop haptics are also designed to optimize positional stability of the CT LUCIA 621P.
As a fully preloaded lens, the CT LUCIA 621P has safety benefits, regardless of the surgical setting, because it eliminates the potential for IOL contamination and damage that can occur with manual loading. The fully preloaded injector system also enables surgical efficiency as it is a surgeon-friendly device that consistently delivers the lens smoothly into the capsular bag through a 2.2 to 2.6 mm incision (depending on IOL power). Once released into the eye, the CT LUCIA 621P unfolds gently thanks in part to its heparin-coated surface*, and the controlled unfolding behavior of the lens facilitates its intraocular positioning.
The CT LUCIA 621P clinical trial results
Our clinical trial investigating outcomes after cataract surgery with the CT LUCIA 621P was a prospective, single center study enrolling patients with clinically significant (Lens Opacities Classification System [LOCS] III grade ≥3) senile cataract. Eligible patients were adults deemed suitable for undergoing extracapsular cataract extraction via phacoemulsification followed by implantation of a posterior chamber IOL with a calculated power between +4.0 D and +34.0 D (in 0.5 D increments). In addition, patients were included only if they were anticipated to attend follow-up examinations scheduled at postoperative months 1 and 3 and had the potential to achieve corrected distance visual acuity (CDVA) 0.3 logMAR or better. Biometry measurements were obtained with the IOLMaster 700 or with A-Scan if cataract density precluded use of swept-source optical coherence tomography technology.
We enrolled 81 eyes of 52 patients and remarkably had follow-up data for 79 eyes. The patients had a mean age of 68.3 years, and females represented a large majority. Preoperatively, logMAR CDVA was ≥0.3 in 38 eyes (47%).
Monocular CDVA ≥0.3 logMAR or better at 3 months was the primary endpoint and was achieved by 79 eyes (100%). This perfect result surpasses the aforementioned WHO target for visual outcomes after cataract surgery set and also significantly exceeds the ISO Safety and Performance Endpoint for outcomes in clinical investigations of IOLs requiring 92.5% of eyes achieve logMAR CDVA ≥0.3(P < 001).1,6 Impressively at 3 months, logMAR CDVA averaged 0.05±0.07 and was 0.1 (20/25) or better in 67 eyes (85%) and 0.0 (20/20) or better in 46 eyes (58%).
We plan to report additional outcomes from this study in a manuscript that will be submitted for publication in a peer-reviewed journal. However, it is worth mentioning that protocol-specified procedures at postoperative visits included examination for IOL glistenings and PCO. At 3 months, neither of those issues were identified in any eye nor did we observe any cases of significant IOL decentration.
Conclusion
Obtaining excellent results after cataract surgery in developing countries is important considering that access to future ophthalmic care can be poor. However, achieving this goal can be challenging because of a variety of patient- and infrastructure-related issues. Our study enrolled patients with relatively dense cataracts who may have had unidentified vision-limiting pathology and poor compliance with postoperative instructions. Yet, the CDVA outcomes (average 0.05±0.07 logMAR) in our study are on par with those reported in rigorous premarketing and postmarketing clinical trials conducted in Western countries. We believe that use of the IOLMaster 700 and CT LUCIA 621P contributed to our impressive functional results in a difficult setting and at the same time provided advantages for streamlining workflow and increasing safety.
*Fragment of heparin used in IOL surface coating with no pharmacological, immunological or metabolic action.
References
- World Health Organization. Programme for the Prevention of Blindness and Deafness. Informal Consultation on Analysis of Blindness Prevention Outcomes. February, 1998.
- Borkenstein AF, Borkenstein EM. Geometry of acrylic, hydrophobic IOLs and changes in haptic-capsular bag relationship according to compression and different well diameters: a bench study using computed tomography. Ophthalmol Ther. 2022;11(2):711-727.
- For more details on ZEISS OPTICS [ZO] asphericity concept, please see:
Borkenstein AF. Cataract surgery in challenging cases – choosing the CT LUCIA 621P IOL to optimize success in eyes with compromised zonules. Ophthalmology Times Europe. 2021 July/August Vol .17 No.6 - Maedel S, Evans JR, Harrer-Seely A, Findl O. Intraocular lens optic edge design for the prevention of posterior capsule opacification after cataract surgery. Cochrane Database Syst Rev. 2021;8(8):CD012516.
- Donachie PHJ, Barnes BL, Olaitan M, Sparrow JM, Buchan JC. The Royal College of Ophthalmologists' National Ophthalmology Database study of cataract surgery: Report 9, Risk factors for posterior capsule opacification. Eye (Lond). 2022 Aug 24. Epub ahead of print.
- ISO 11979-7:2018. Ophthalmic implants — Intraocular lenses — Part 7: Clinical investigations of intraocular lenses for the correction of aphakia.
Dr Kretz is Managing Director at the Precise Vision Augenärzte, Headquarter, Rheine, Germany and CEO of the German NGO Augenärzte für die Welt gGmbH, Rheine, Germany
Professor Shah is a Consultant Ophthalmic Surgeon at the Birmingham and Midland Eye Centre, Birmingham, UK
Dr Kretz is a paid consultant to Carl Zeiss Meditec.
Newsletter
Get the essential updates shaping the future of pharma manufacturing and compliance—subscribe today to Pharmaceutical Technology and never miss a breakthrough.



























